[ad_1]
Experiments results detailed in Table 2 for understanding the impacts of KMnO4 in ECDD on M.S sheets, focusing on hole dimensional accuracy in terms of over/undercut (%), MRR, and TWR. Tool wear, measured by tool electrode length reduction percentage, was also analyzed. The complex material removal process in ECDD has been theorized by various researchers.
One study suggests metal removal occurs through electrical sparks generated during the switching phenomenon rather than by breaking down the insulating gas layer50. Conversely, another researcher posits a mechanism involving local heating and chemical etching51. Additionally, the arc discharge valve theory proposes a mechanism for metal removal from the workpiece52. From this investigation, it appears that metal removal primarily results from localized workpiece melting due to heat penetration from sparks, involving gas film discharge, spark and chemical erosion, and atom-by-atom removal. The stability of the gas film, particularly during discharge and hydrodynamic regimes, significantly enhances ECDD performance, notably with the use of a 5% KMnO4 mixed NaOH electrolyte. According to Fig. 3, the study evaluated the hole diameter i.e. % of Overcut/undercut, MRR, TWR, and tool length reduction under different electrolyte combinations: plain NaOH (7.5 M), 5% KMnO4 mixed with NaOH (7.22 M), 50% NaOH and 50% KOH (5.36 M), and 5% KMnO4 mixed with 47.5% NaOH and 47.5% KOH. Each data point in Fig. 3 corresponds to the average of three independent experimental trials. The associated error was carefully evaluated and found to be within ± 5%.
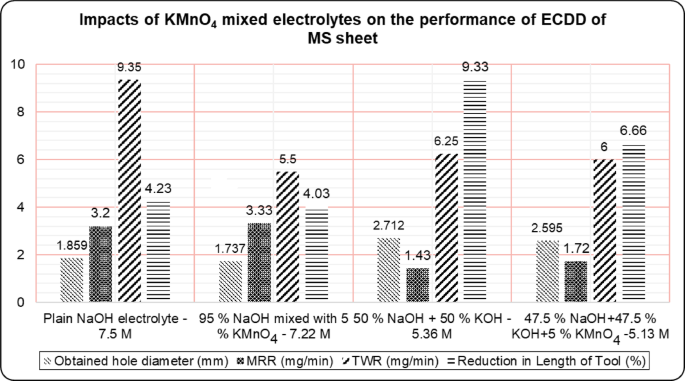
Impacts of KMnO4 mixed electrolytes on the performance of drilling in the ECDD of MS sheet.
Effects of KMnO4 on dimensional accuracy of obtained drilled hole
This study aimed to achieve high-dimensional accuracy of hole in the ECDD process. This task is particularly challenging due to issues such as stray current erosion resulting in entry hole having a larger diameter than the inner part. Although the tool is insulated, it degrades due to high-intensity sparks generated during the ECDD process. Factors like bubble generation, gas film characteristics, the potential difference between the tool and auxiliary electrodes, and electrolyte conductivity significantly influence the quality of the holes produced in ECDD. To enhance accuracy, KMnO4, an oxidizing agent, was mixed with alkaline electrolytes (NaOH and KOH). To achieve precise holes, various electrolyte combinations were tested: plain NaOH, 95% NaOH with 5% KMnO4, 50% NaOH and 50% KOH, and 47.5% NaOH and KOH each with 5% KMnO4. Based on the results of the preliminary experiments, a maximum of 5% (wt.) KMnO4 was chosen for the above combinations of electrolytes.
Basically, KMnOâ is a potent oxidizing agent that can facilitate the breakdown of passivating layers that form on the workpiece surface, such as oxides or other insulating films. In the ECDD process, the presence of KMnOâ in the electrolyte accelerates the oxidation of these layers, preventing them from passivating the surface. This maintains the electrical conductivity of the machining gap and ensures a consistent MRR. When KMnOâ is mixed with other electrolytes like NaOH/KOH, it undergoes reduction at the cathode while simultaneously causing the oxidation of metallic ions at the anode. The reactions between KMnOâ and these bases create a more aggressive electrolytic environment. The reduction of MnOââ» to MnOâ in alkaline conditions results in the release of oxygen gas, which helps in maintaining a stable gas film around the tool electrode, enhancing the electrochemical discharge stability.
The mixed electrolyteâs ability to maintain a consistent current density across the machining gap helps in achieving better control over the electrochemical reactions. This prevents the formation of excessive heat and localized damage, minimizing the HAZs and enhancing the dimensional accuracy of the drilled holes. The novel electrolyte combination is specifically designed to optimize the balance between oxidative and reductive reactions, ensuring improved material removal, reduced passivation, and consistent drilling performance. Moreover, KMnO4 helps to enhance the thermal and electrical stability of the electrolytes, providing a stable drilling environment by preventing fluctuations in electrolyte properties. Figure 4 presents the optical images of the holes obtained using different electrolytes.
The recorded hole sizes were: 106.23% of the tool diameter with plain NaOH, 99.26% with 5% KMnO4 and 95% NaOH, 154.97% with NaOH and KOH, and 148.29% with 5% KMnO4, 47.5% NaOH, and 47.5% KOH against the tool electrode diameter of 1.75Â mm. The use of KMnO4 mixed with NaOH resulted in a hole diameter of 1.737Â mm, deviating by only 0.74% from the 1.75Â mm tool electrode diameter. Additionally, combining NaOH and KOH with KMnO4 yielded the hole diameter of 2.595Â mm, compared to 2.712Â mm with plain NaOH and KOH, showing a 4.5% higher overcut. The combination of KMnO4 with NaOH also controlled stray corrosion by forming a thin layer over the tool surface during ECDD and prevented localized heating, resulting in dimensional accuracy. It is concluded that the addition of KMnO4 significantly improves the accuracy of holes in ECDD, meeting industrial demands for precise drilling in advanced engineering materials with high hardness, brittleness, and poor conductivity.

Optical microscope images of obtained holes using different electrolytes. (a) Plain NaOH electrolyte (b) 95% NaOH mixed with 5% KMnO (c) 50% NaOH + 50% KOH (d) 47.5%NaOH+â47.5% KOHâ+â5% KMnO4
Effects of KMnO4 on MRR and TWR
The introduction of KMnOâ, a strong oxidizing agent, modifies the local electrochemical environment by increasing the oxygen content in the machining gap, which facilitates the dissolution of metal ions from the workpiece surface. The presence of KMnOâ in an alkaline medium, such as NaOH or KOH, prevents the formation of passivating oxide layers that can impede the metal removal process, thereby enhancing the overall material removal mechanism. In an alkaline solution like NaOH, KMnOâ can undergo redox reactions that help stabilize the electrolyte, which in turn contributes to a more consistent and controlled drilling process. This stability reduces fluctuations in current density, improving the uniformity of the electrochemical discharges and enhancing the precision and quality of the machined holes. The reaction of KMnO4 in an alkaline medium can be represented as presented in Eq. 253. Here, KMnO4 gets reduced to Na2MnO4, and this helps in maintaining the oxidative environment necessary for the ECDD process.
$$\:2KMn{O}_ generator installation+2{H}_ electrical installationO+3NaOH\underrightarrow{}2KOH+2{Na}_ electrical installationMn{O}_ generator installation+2{H}_ electrical installation{O}_ electrical installation$$
(2)
The high pH of NaOH ensures the generation of hydroxide ions (OHâ»), which are essential for maintaining conductivity and facilitating chemical reactions during the drilling process. MRR generally depends on the applied voltage between the tool electrode and the auxiliary electrode, the thermal and electrical conductivity of the electrolytes, the stability of electrolytes during spark discharge, the pH value, and the formation of hydrogen and oxygen gas films due to the use of KMnO4. Among the four electrolyte combinations tested, results revealed that KMnO4 significantly improves the MRR. A higher MRR of 3.33Â mg/min was achieved using 95% NaOH with 5% KMnO4, which is 4.1% higher than plain NaOH, 132.87% higher than the 50% NaOHâ+â50% KOH combination, and 93.6% higher than using 47.5% NaOHâ+â47.5% KOHâ+â5% KMnO4 electrolytes. Despite the higher pH value of plain NaOH, the stability of the electrolyte was better maintained when using KMnO4, resulting in a higher MRR than any other electrolyte combinations.
In this study, the improved tool life observed with KMnOâ addition is attributed to its strong oxidizing nature, which is likely to facilitate the formation of a thin protective layer on the tool electrode surface. While direct experimental confirmation of this layer is not available in the present work, the performance trends summarized in Table 2 support this probable mechanism. The texture of the electrode surface plays a crucial role in bubble generation and coalescence, which are key factors in ensuring better MRR and TWR in ECDD. Generally, stray corrosion, instability in electrolyte conductivity, and spark intensity directly affect tool life. Therefore, the dimensional accuracy of the hole depends on the dimensional stability of the tool electrode during the ECDD process. The lowest TWR obtained was 5.5 mg/min using the 95% NaOHâ+â5% KMnO4 electrolyte, which is 70% lower compared to plain NaOH. This provides concrete evidence of KMnO4âs impact on achieving minimal TWR, as represented in Fig. 3.
The combination of NaOH and KOH did not significantly achieve the chosen lower TWR. Plain NaOH led to higher tool wear and notable tool electrode erosion despite NaOH and KOH being severe alkaline electrolytes with good ionic conductivities. Consequently, better MRR can be obtained by sacrificing tool electrode life. It is concluded that tool electrode life is reasonably improved when using KMnO4 mixed with NaOH electrolytes due to the formation of a protective thin layer on the tool electrode surface, making it suitable for ECDD applications. The percentage of reduction in tool length is also presented in Fig. 3. The experimental results revealed that the highest reduction in tool length occurred when using the 50% NaOHâ+â50% KOH electrolyte, followed by plain NaOH. The lowest reduction in tool length (4.03%) was observed when using the 95% NaOHâ+â5% KMnO4 electrolyte for making holes in ECDD, which is 132% lower.
Effects of KMnO4 on HAZ
The HAZ was identified as the peripheral ring-like region surrounding the hole, where microstructural changes due to thermal exposure were evident as shown in Fig. 5. The HAZ analysis was carried out using Scanning electron microscope (SEM) images for different electrolyte conditions: (a) Plain NaOH (b) 95% NaOHâ+â5% KMnOâ (c) 50% NaOHâ+â50% KOH (d) 47.5% NaOHâ+â47.5% KOHâ+â5% KMnOâ. The scale bar (200 μm) indicates that the average HAZ width is in the range of ~â100â250 μm around the periphery. From the qualitative SEM analysis, the results have been identified:
-
Plain NaOH (a): A wide and irregular HAZ was observed, indicating higher thermal damage and unstable discharge conditions.
-
95% NaOHâ+â5% KMnOâ (b): The narrowest and most uniform HAZ was observed in this condition, suggesting the beneficial role of KMnOâ in stabilizing discharge and reducing excess heating.
-
50% NaOHâ+â50% KOH (c): The HAZ appeared moderately reduced compared to plain NaOH, reflecting improved ionic conductivity and controlled discharge spread.
-
47.5% NaOHâ+â47.5% KOHâ+â5% KMnOâ (d): A comparatively narrower HAZ was seen in this condition, highlighting the effect of mixed electrolytes with KMnOâ in minimizing thermal damage.
Generally, the use of alkaline electrolytes such as NaOH and KOH enhances MRR but sacrifices material properties at the entrance area of the electrochemically drilled workpieces. This effect is also seen with the combination of NaOH and KOH electrolytes, as evidenced by the SEM images in Fig. 5c. The HAZ is greater when using plain NaOH and combined NaOH and KOH electrolytes54. However, the use of a KMnO4 mixed NaOH electrolyte solution results in better hole size and MRR with minimal HAZ.
The KMnO4 reacts with alkene bonds, shifting electrons and creating bonds to form a cyclic manganese compound. In the next stage, NaOH enters the reaction and hydrolyzes the bonds to form a diol under cold conditions. However, during continuous spark generation in the machining gap, this reaction leads instead to the formation of a carbonyl group. In ECDD, the interaction between KMnOâ and NaOH electrolytes is crucial, particularly for the HAZ. When KMnOâ is mixed with NaOH, it acts as a strong oxidizing agent, forming a more stable and conductive electrolyte. This stability ensures consistent electrical discharges, leading to more precise material removal. The oxidizing properties of KMnOâ may also contribute to the formation of a protective layer on the tool electrode, which in turn can reduce tool wear. Additionally, KMnOâ presence improves breakdown voltage, enhancing the sparking mechanism. This controlled sparking reduces thermal damage to the workpiece, thereby minimizing the HAZ. Consequently, localized heating is confined to a smaller area, improving the dimensional accuracy of the drilled features and reducing unwanted thermal effects like micro-cracking and surface oxidation. Overall, using KMnOâ mixed with NaOH electrolyte improves the ECDD process in terms of MRR and TWR, significantly reducing adverse impacts on the HAZ and leading to better dimensional accuracy with minimal HAZ. The presence of carbonyl groups enhances heat dissipation from the tool surface.
It is well established that KMnOâ oxidation can introduce carbonyl and other oxygenated functional groups on surfaces, which in turn improve electron mobility and thermal conductivity55. In the present experiments, the observed reduction in tool wear is attributed to the probable formation of carbonyl groups on the tool electrode surface during the ECDD process. This arises from the oxidative interaction of KMnOâ with organic residues or hydrocarbon species generated under spark discharge conditions. Under continuous sparking in an alkaline environment, KMnOâ tends to oxidize unsaturated bonds, leading to the formation of carbonyl functionalities such as aldehydes and ketones on the electrode surface or at the electrolyteâelectrode interface. These polar groups are likely to enhance thermal conductivity and promote more efficient heat dissipation from the tool surface, thereby reducing localized overheating and minimizing tool wear.
Such surface chemical modifications have been demonstrated to increase surface energy and thermal stability, as reported in studies on electrochemical surface oxidation and modification. Carbonyl groups improve electron mobility and contribute to more uniform and stable sparking by stabilizing the electrolyte-electrode interface, which in turn reduces erratic tool wear caused by inconsistent discharge intensities56. Efficient heat dissipation reduces thermal stress and prevents the tool from overheating, which is one of the key factors in minimizing tool wear during the ECDD process. Carbonyl groups can also influence the sparking process by creating a more uniform and stable discharge environment. This uniformity minimizes the intensity and irregularity of electrical sparks, thereby reducing erratic tool wear caused by inconsistent sparking. Consequently, the use of KMnO4 with alkaline electrolytes, specifically NaOH, improves the lifespan and geometrical stability of the tool, enhancing the overall performance of the ECDD process.
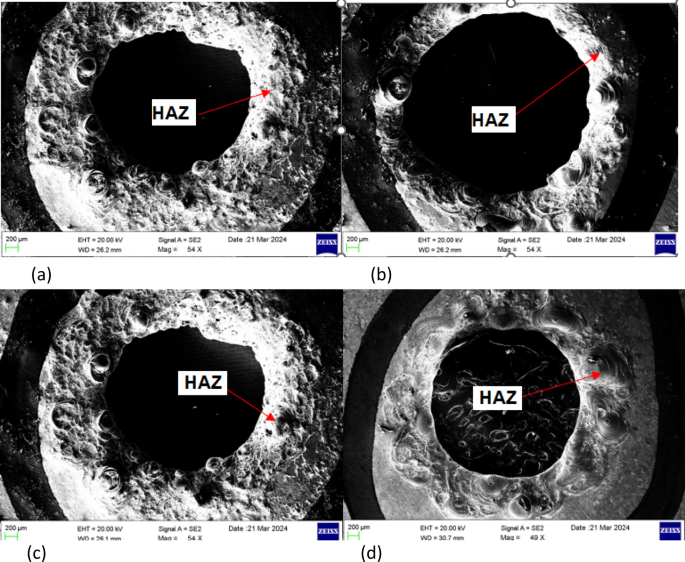
SEM images of holes with the heat affected zones while using different electrolytes with/without KMnO4. (a) Plain NaOH electrolyte (b) 95 % NaOH mixed with 5 % KMnO4 (c) 50 % NaOH + 50 % KOH (d) 47.5 % NaOH+47.5 % KOH+5 % KMnO4
Potential limitations of using KMnOâ with 95% NaOH in industrial applications
While the addition of 5% KMnOâ to 95% NaOH electrolyte offers significant improvements in ECDD performance, certain limitations and challenges must be considered for broader industrial adoption. Firstly, the chemical stability of KMnOâ in highly alkaline solutions can be compromised over extended use due to its strong oxidizing nature, potentially leading to gradual depletion of active species and requiring frequent electrolyte replacement or replenishment to maintain machining consistency.
Secondly, electrode contamination is a concern as KMnOâ and its reduction products, such as manganese dioxide (MnOâ) can deposit on the tool electrode surface or workpiece, potentially affecting electrical conductivity and the uniformity of spark discharges. This buildup may necessitate periodic cleaning or additional filtration systems in the electrolyte circulation loop.
Furthermore, the intensity of electrical discharges can be influenced by the electrolyte composition; strong oxidizers like KMnOâ would intensify sparking, if not properly controlled, can lead to uneven wear, micro-cracks, or unwanted surface alterations, thus requiring precise parameter optimization and process monitoring.
Safety and environmental considerations must also be addressed. KMnOâ is a hazardous oxidizing chemical, presenting risks such as chemical burns, toxicity, and environmental harm if mishandled or improperly disposed of. Industrial operations must implement strict safety protocols and ensure responsible waste management to mitigate these risks. Additionally, handling highly concentrated NaOH demands caution due to its corrosive nature.
Overall, while KMnOâ mixed with 95% NaOH demonstrates clear process benefits, these limitations highlight the need for further research into electrolyte stability, system design for contamination control, spark intensity regulation, and adherence to safety and environmental standards to enable safe, sustainable, and efficient industrial usage.


